 It is important to understand that controls-either positive or negative-can never prove the identity-or the presence or absence-of a molecule in a tissue sample using immunohistochemical techniques. The possibility of both errors must be considered when designing an immunohistochemical assay. Both can lead to erroneous scientific conclusions and clinical misdiagnoses. It’s equally important not to conclude that a molecule is present when in fact it is not, as it is to conclude that it is absent when in fact it is present. Simply stated, an immunohistochemical assay that lacks controls cannot be validly interpreted. 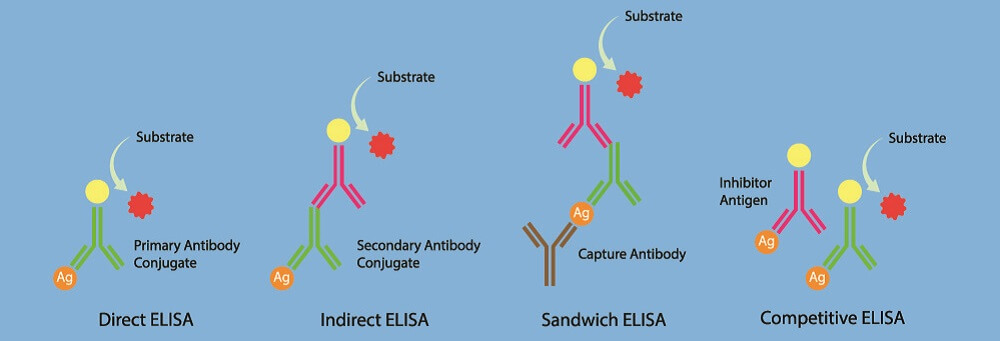
Furthermore, controls must be described and included in the ensuing publications. Realizing that controls are an essential component of experimental design in all scientific investigations, we insist that valid interpretations of immunohistochemical assays cannot be made in the absence of minimally appropriate controls. Contributing to this problem is the fact that many high-impact journals that publish immunohistochemical data do not require authors to report controls for immunohistochemistry and, what is perhaps more regrettable, the controls that are published often are inappropriate or misinterpreted. This neglect has resulted in the publication of unverified and irreproducible findings in the literature ( Couchman 2014 Collins and Tabak 2014). 2013), it is the experience of the authors that critical controls are often not performed or reported in ensuing publications. 
Unfortunately, in the case of immunohistochemistry, although the need for controls is well established ( Baskin 2009 Burry 2000, 2010 2011 Frevert et al. The sine qua non of validating research findings is the use of appropriate controls in the design and performance of experiments and assays. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
